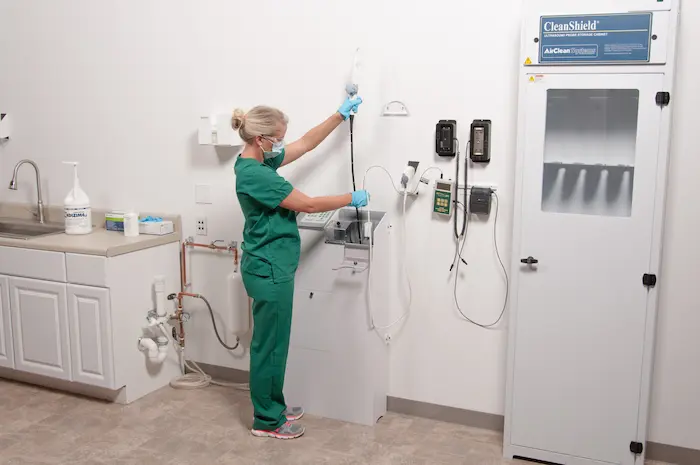
The TD 100 delivers high level disinfection to TEE probes in ONLY 5 minutes. The TD 100 disinfects each TEE probe with High-level disinfectant and provides printed verification upon completion of successful disinfection cycle. Microprocessor-controlled, the TD 100 is simple to use for any health care professional reprocessing a used TEE ultrasound probe.
The TD 100 dramatically reduces the amount of stress and the potential for damage to TEE probes. The system was designed with every aspect of the TEE probe taken into consideration. The control handset is held securely within the TD 100, while strain and stress is minimized from the cable and electronic pack adapter with a specially designed hanger and mounts for both.
Additionally, the height to insert the TEE probe distal tip and shaft is only 38", giving less potential of hitting and damaging the distal tip during insertion into the TD 100.
The TD 100 suspends the TEE probe in a custom fitted bracket that holds the control handle securely while the insertion shaft of the probe is placed within the disinfection reservoir for high-level disinfection. The TD 100 is designed to prevent oversoaking as this is the second leading reported damage to TEE probes beyond being dropped or struck by another object. Manual reprocessing can expose your TEE probes to this type of expensive damage.
The following is a manufacturer’s warning for TEE probes:
Do not use a transducer that has been dropped or struck against another object. The transducer is fragile and will break ... this is not covered by the probe warranty or service contract.