Introduction
CS Medical LLC agrees with the CDC and recommends high-level disinfection is followed by rinsing and drying1 The current commonly used methods of drying ultrasound probes, such as with a cotton cloth, a polyester fabric, or a paper towel, can potentially result in bacterial contamination. For the reason of patient safety, we sought to ascertain if common ultrasound probe drying materials were contaminated with microbes. This is an important issue because of the high prevalence of microbial contamination of ultrasound transducers: 12.9% of transducers are contaminated with pathogenic bacteria following disinfection2, up to 7.5% of transvaginal ultrasound transducers were found to have cancer-associated HPV DNA after low-level disinfection with wipes3, and rates of probe cover perforation of ultrasound endovaginal probe covers have been measured to be as high as 9%4• Failure with regards to these types of infections are potentially most devastating to immuno-compromised patients. The experimentation reported here tests a potential "weak" link in the "chain" of high-level disinfection custody" of ultrasound probes.
Summary
A standard bacteriological analysis was conducted to determine the inherent bioburden on three materials that are commonly used to dry ultrasound probes: cotton cloth, polyester fabric, and paper towels. The results of this analysis showed that all three materials were contaminated with bacteria. This finding shows that these common methods of wiping dry ultrasound probes should be re-evaluated for patient safety. The results of this analysis suggest that a straight-forward way to reduce microbial contamination of ultrasound transducers is to perform the post-disinfection drying step with a sterile or gamma irradiated cloth.
Method of Analysis
Samples of paper towels, cotton cloths, and polyester fabric were immersed in freshly made nutrient broth to encourage growth of any present bacterial cells. A positive control was used to show performance efficacy of media, and a negative control was used to show sterility of media. The negative and positive control passed as expected. The negative control plate demonstrated no growth of colony forming units, and the positive control plate demonstrated growth of colony forming units.
Results and Conclusion
All paper towel samples, taken from three different locations, each tested in duplicate, showed inherent bioburden is present. All polyester fabric samples, and cotton cloth samples, tested in duplicate, also showed the presence of inherent bioburden. The photographic evidence in the appendix shows bacterial growth present on all plates, excluding the negative control.
Copyright 2018. All rights reserved. No portion of this document may be reproduced without written permission from CS Medical, LLC.
Conclusion
Our testing shows that common drying methods for ultrasound probes can potentially increase the risk of bacterial transmission to the patient. The materials tested (cotton, paper towels, and polyester) were found to contain significant bioburden. For increased patient safety in clinical practices, CS Medical heeds the recommendation of the Joint Commission5 for healthcare facility accreditation and certification to "reduce the risk of infection associated with medical devices" by suggesting that users perform post-disinfection ultrasound probe drying with sterile or gamma irradiated products. Our hypothesis is that this straight-forward change to use of a sterile or gamma irradiated drying cloth could prevent the type of bacterial contamination outbreaks that have caused healthcare-acquired infections. This change could eliminate the type of patient microbe transmission problem that has arisen when high-level disinfection is compromised, comparable to when transducer gel or rinse water is contaminatedM6• A reduction in patient infection via ultrasound transducer use is anticipated to be most important for immune-compromised patients. This and other reasons leads us to our conclusion that the drying method for ultrasound probes could be of significant importance for patient health.
References
Rutala Wet al. CDC (2008) Guideline for Disinfection and Sterilization in Health Care: Link
Leroy SJ. Hosp Infect 2013 83(2):99-106.
Ma Set al. Emerg MedJ. 2013 30(6):472-5.
Chalouhi GE, et al.J Gynecol Obstet Biol Reprod (Paris). 2009 38(1):43-50.
Electronically accessed: thejointcommission.org. May 2017. Issue 33. Improperly sterilized or high-level disinfected equipment - a growing problem
CDC (2012) Pseudomonas aeuginosa Respiratory tract infection associated with contaminated ultrasound gel used for transesophageal echocardiography. Morbidity and Mortality Weekly Report, 61 (15) April 20.
Appendix
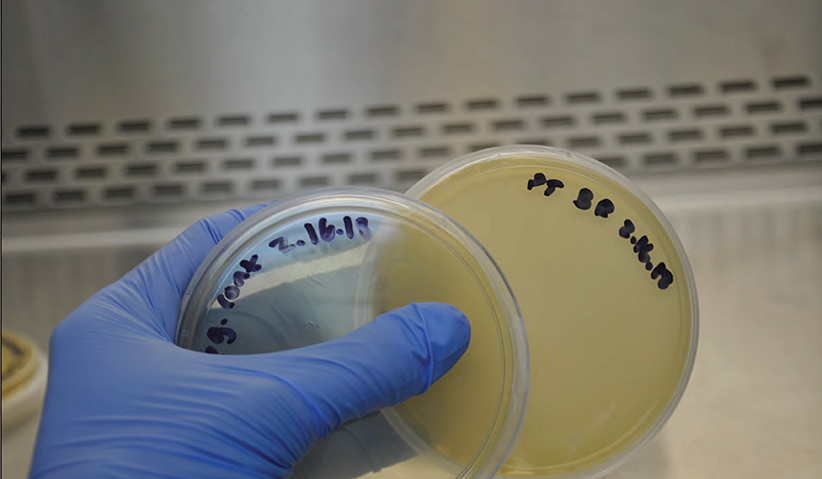
Photo 1: Plates representative of negative control and paper towel from hand sink area.
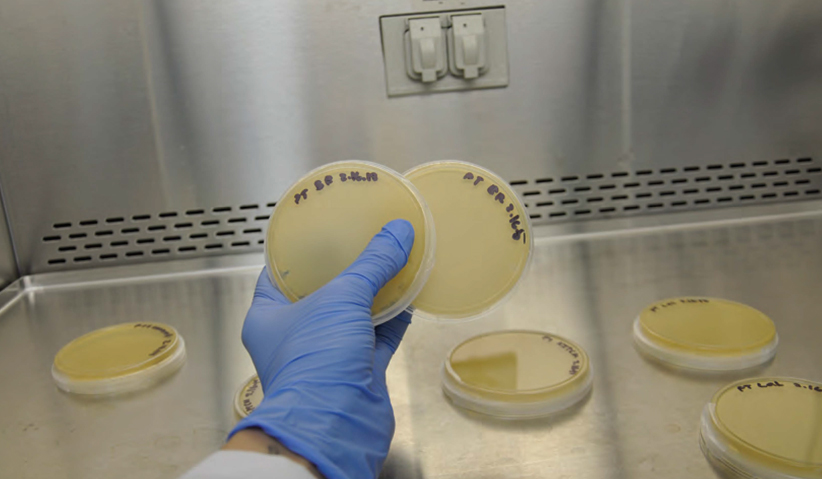
Photo 2: Duplicate plates of paper towel samples from hand sink area, both completely covered with bacteria.
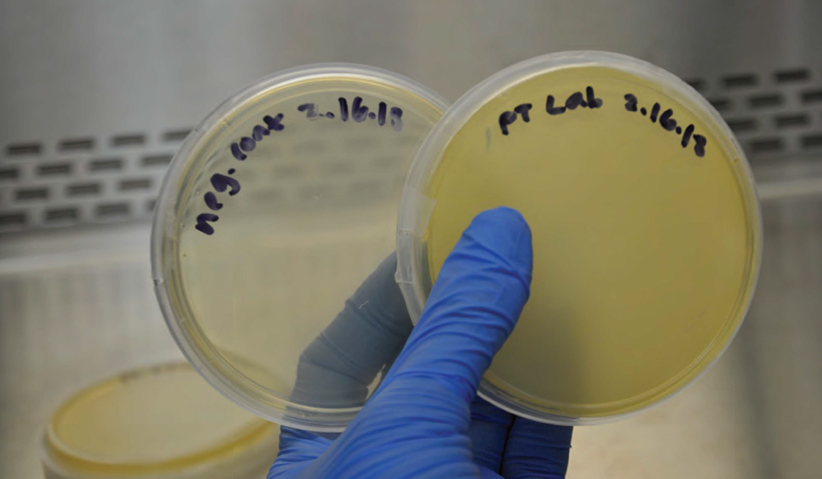
Photo 3: Plates representative of negative control and paper towel from laboratory.
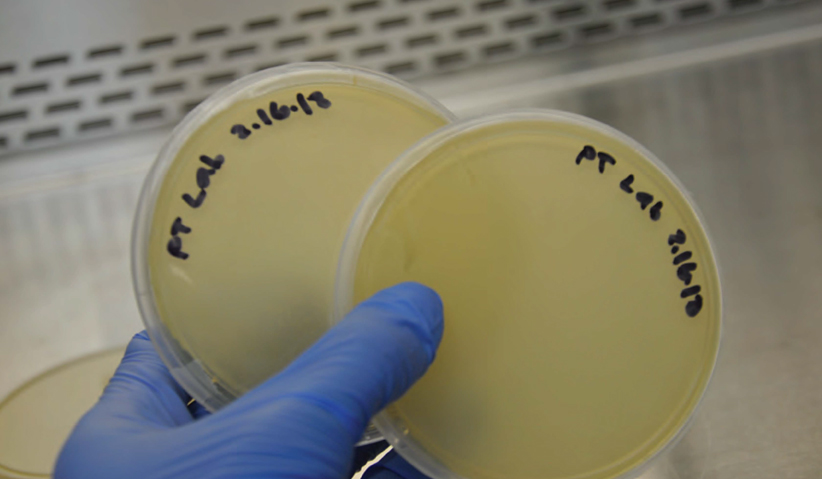
Photo 4: Duplicate plates of paper towel from laboratory, both completely covered with bacteria.
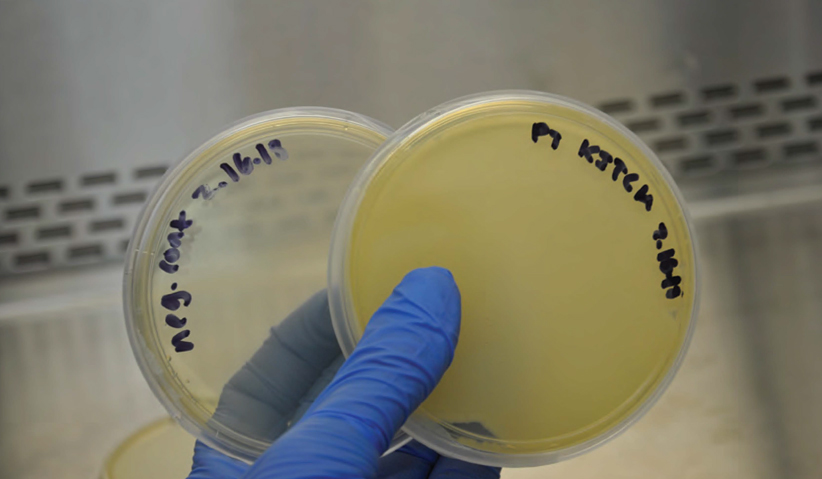
Photo 5: Plates representative of negative control and paper towel from kitchen.
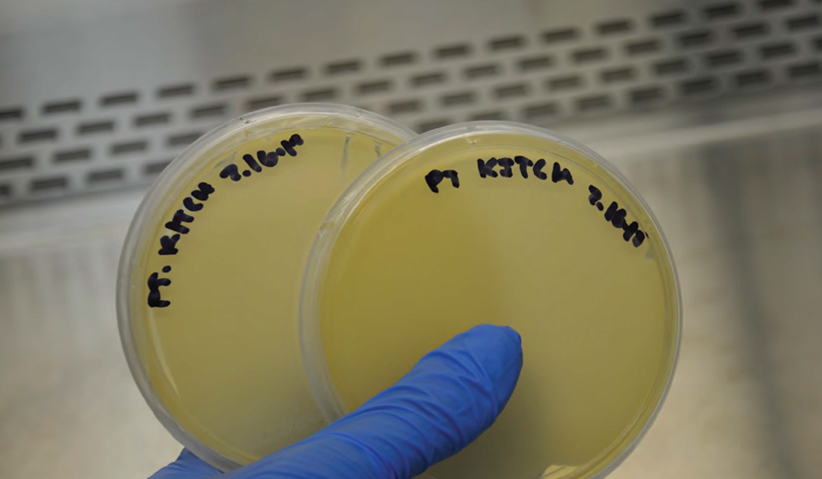
Photo 6: Duplicate plates of paper towel from kitchen, both completely covered with bacteria.
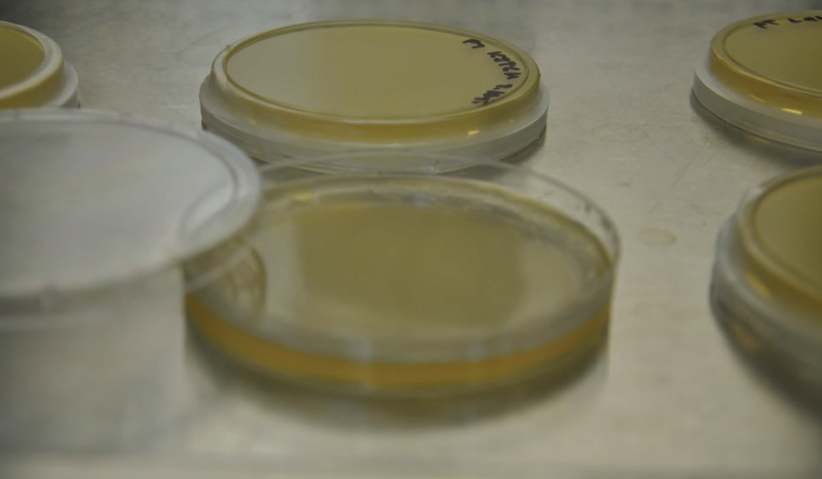
Photo 7: Picture of bacterial covering the agar from a paper towel sample.
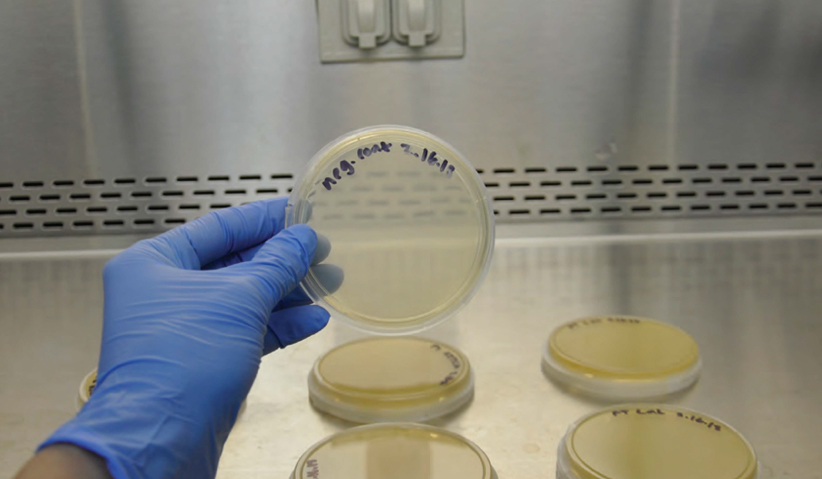
Photo 8: Negative Control exhibits no growth.
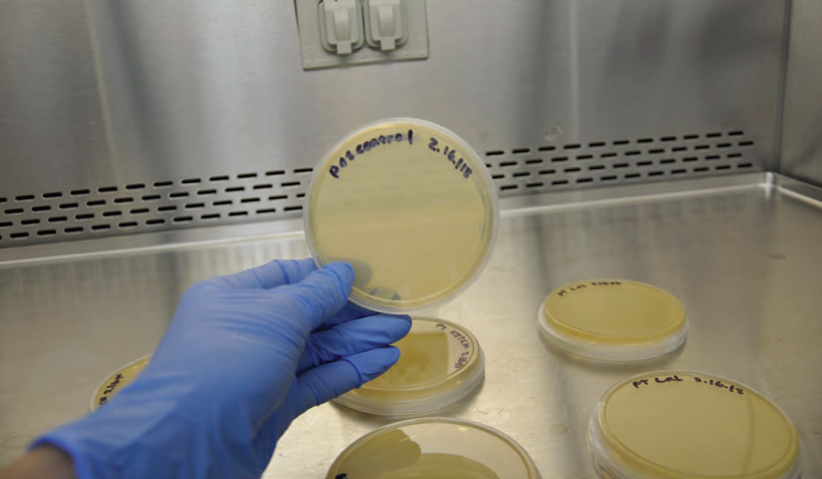
Photo 9: Positive control exhibits growth.
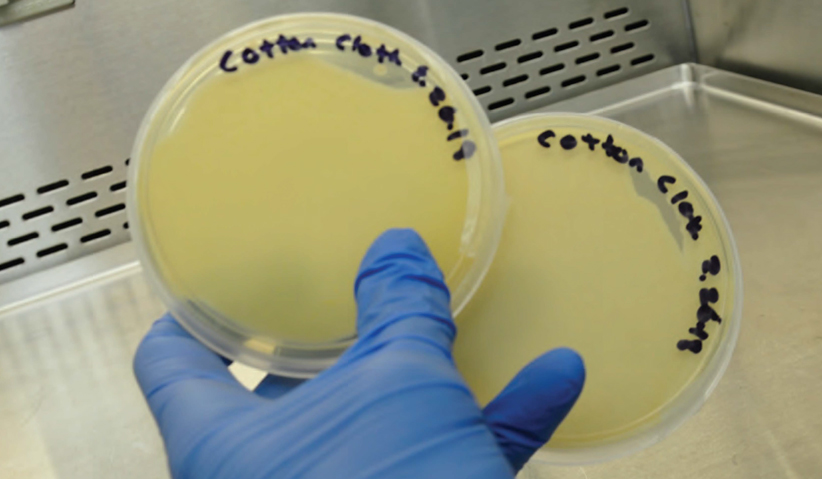
Photo 10: Positive control exhibits growth.
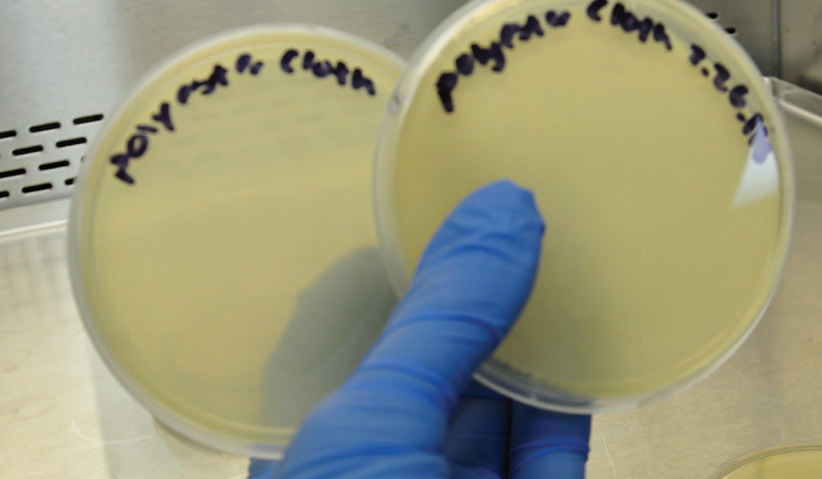
Photo 11: Duplicate plates of polyester cloths, both completely covered with bacteria.

Photo 12: Negative and positive control side by side.

QwikDry® ultrasound drying cloth used in conjunction with CS Medical TD 100® and TEE ultrasound probe.
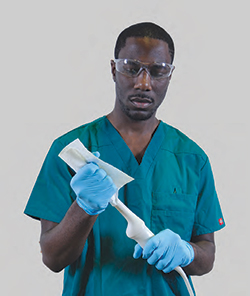
QwikDry® ultrasound drying cloth used in conjunction with transvaginal probe after high-level dinsinfection









