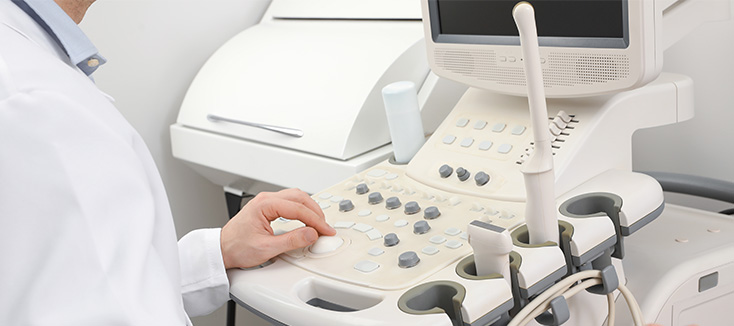
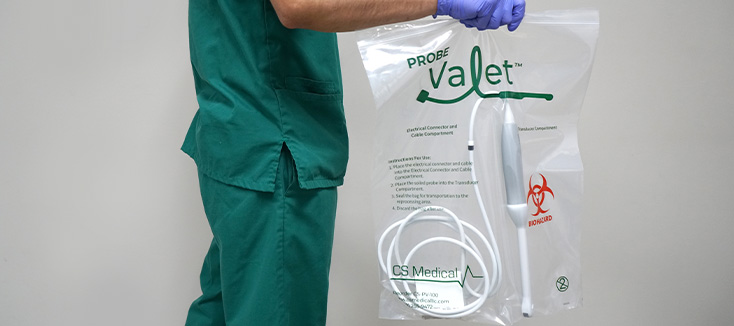
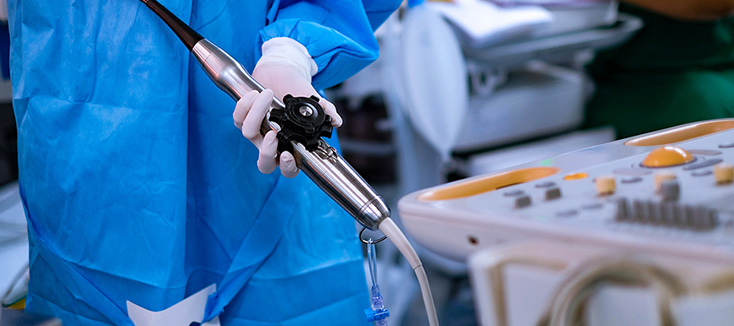
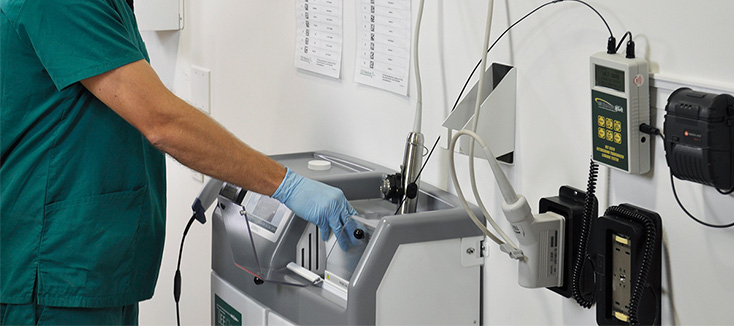
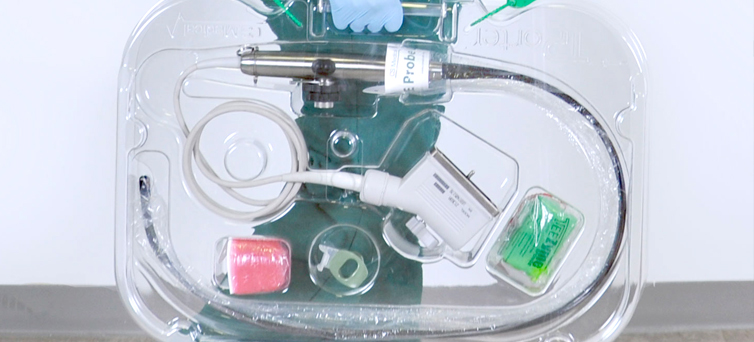
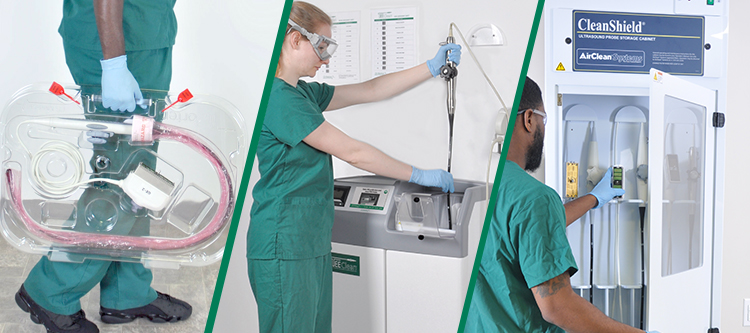
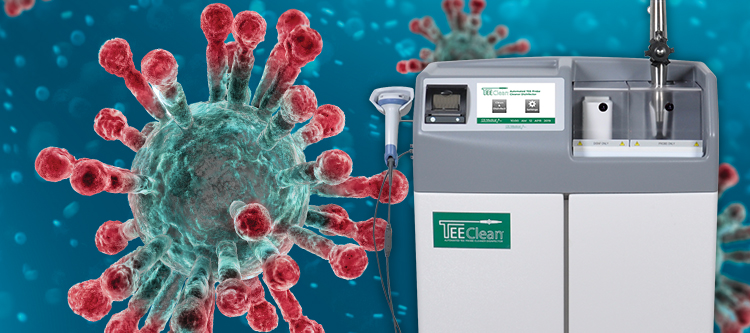
Investing in Patient Safety: CS Medical’s Commitment to IP and SPD Education
Continuing education is vital for healthcare professionals, which is why CS Medical has been committed to providing various avenues for professionals to access free resources. For many facilities, funding for continuing education is among the first items cut during budget tightening. CS Medical recognizes this reality and is responding with action.